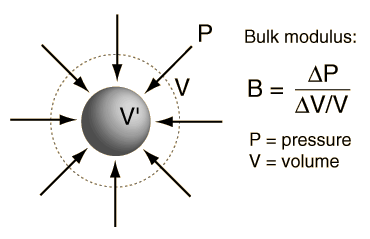
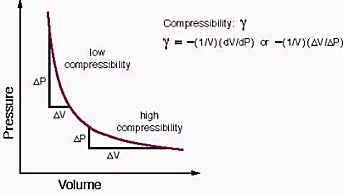
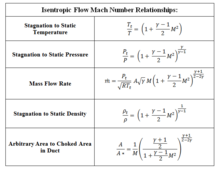
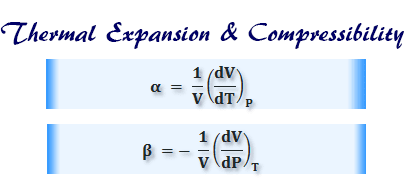SOLVED:Derive an expression for the isothermal compressibility of a substance whose equation of state is P=(R T)/(v-b)-(a)/(v(v+b) T^1 / 2).where a and b are empirical constants.

Developing a Thermodynamical Method for Prediction of Activity Coefficient of TBP Dissolved in Kerosene
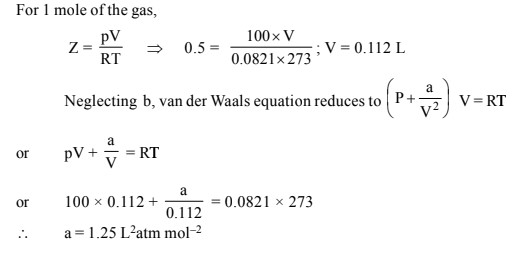
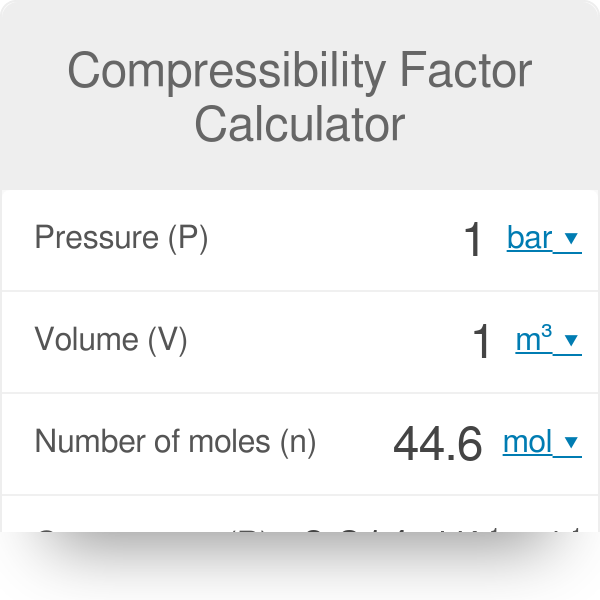
Welcome to Chem Zipper.com......: The compressibility factor for 1 mole of a van der Waals gas at 0oC and 100 atm pressure is found to be 0.5. Assuming that the volume of