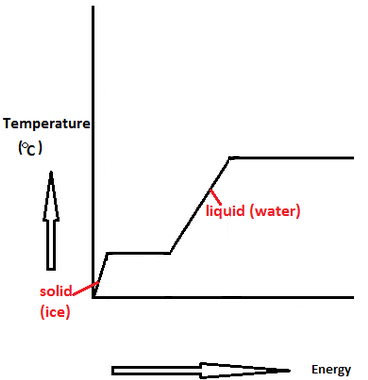
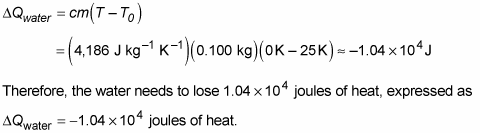SOLVED:Calculate the heat change at 100^∘ C for each of the following and indicate whether heat was absorbed or released: a. calories to vaporize 10.0 g of water b. joules to vaporize

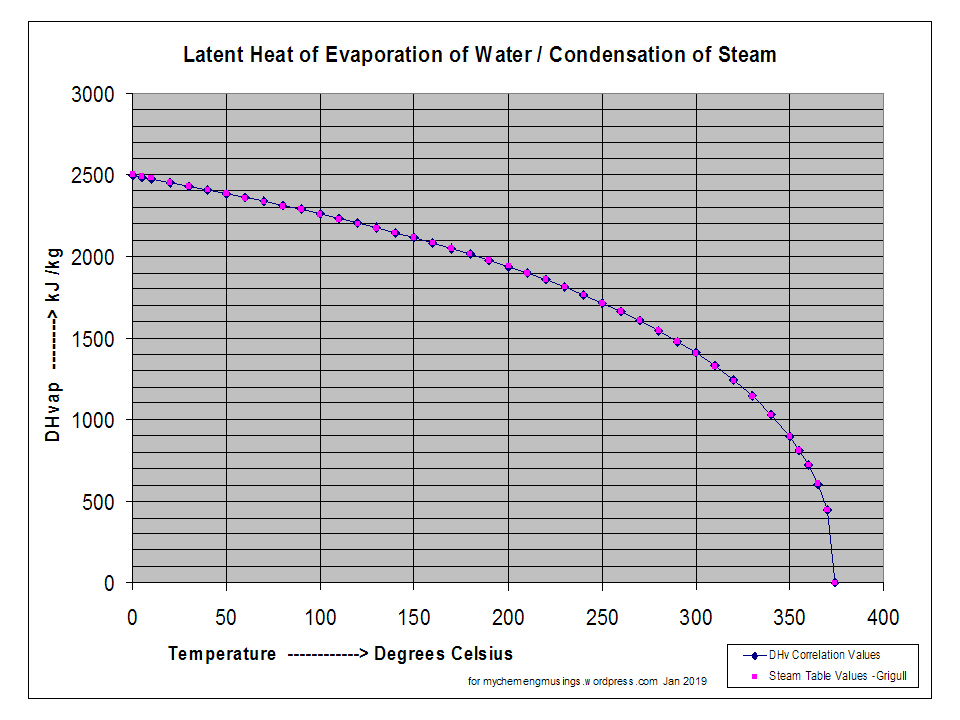
SOLVED: Vapor pressure measured at several different temperature is shown below for mercury- Determine the molar heat of vaporization for mercury with the help of plot T (O 200 250 300 320

Calculate the enthalpy of vaporisation per mole for ethanol. Given, Δ S = 109.8JK^-1mol^-1 and boiling point of ethanol is 78.5^oC .
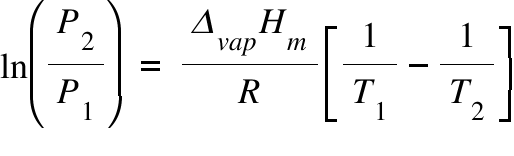
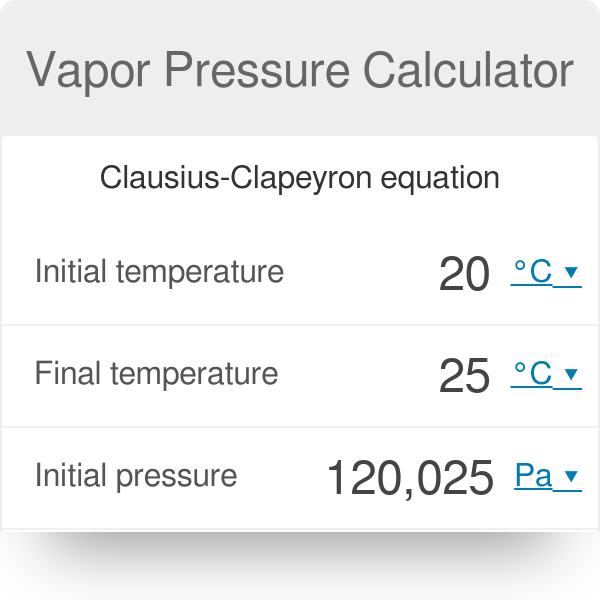
Correlation of Vapor Pressure at Different Temperatures by Clausius Clapeyron Equation Calculator | Calistry

Calculate the entropy change for vaporization of `1mol` of liquid water to stem at `100^()C`, if... - YouTube

Calculate the molal elevation constant, kb for water and the boiling point of 0.1 molal urea solution. Latent heat of vaporisation of water is 9.72 kcal mol ^-1 at 373.15 K.