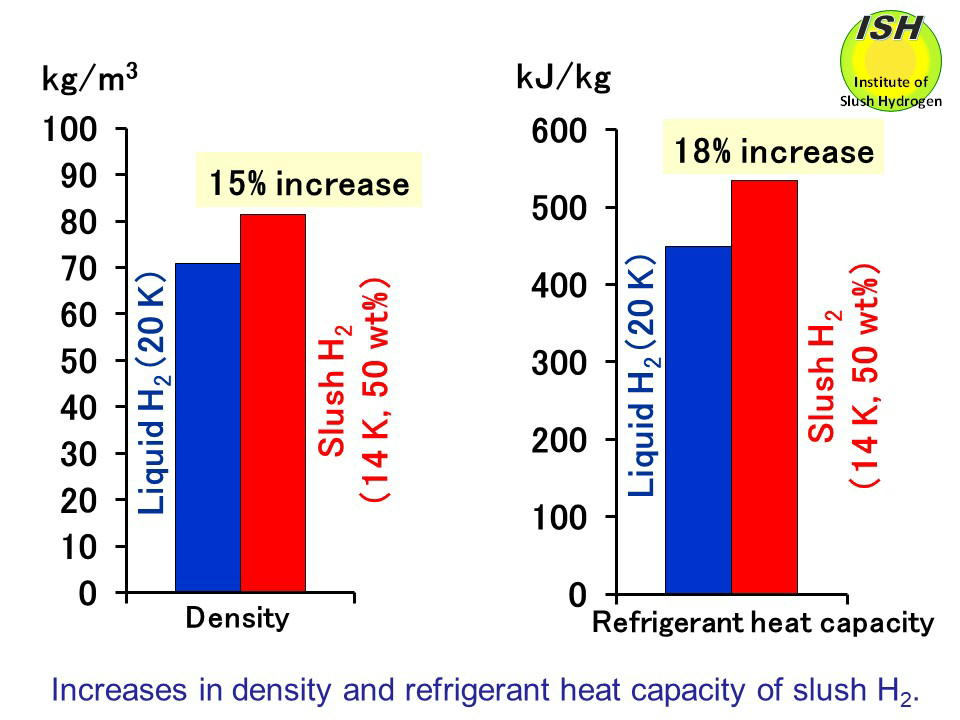
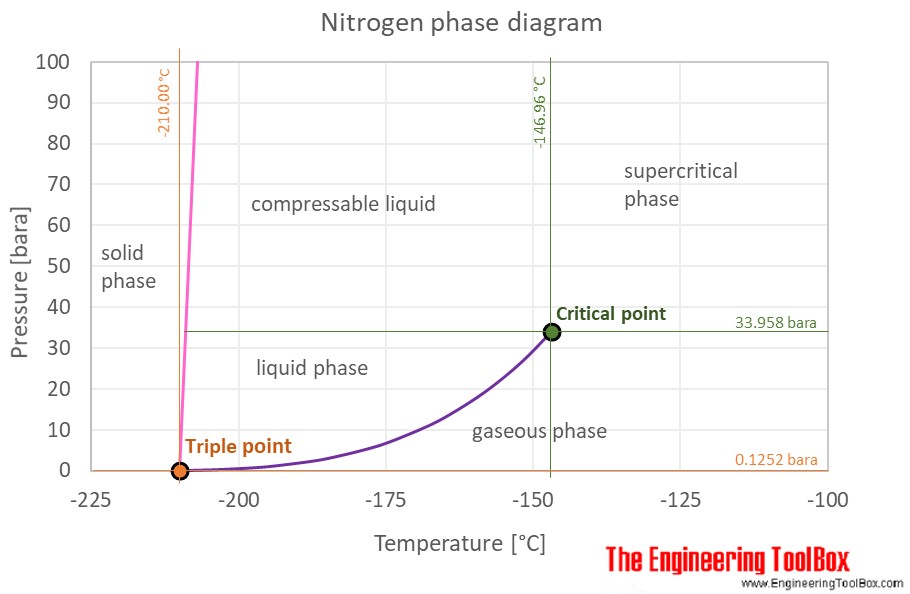
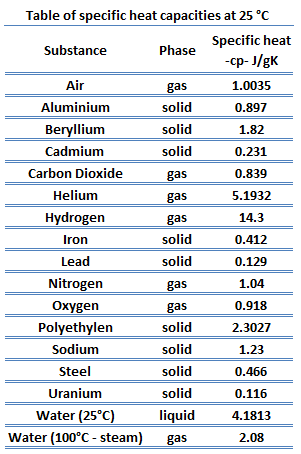
Density and specific heat of nitrogen as function of temperature. Data... | Download Scientific Diagram

The molar heat capacity at constant pressure of nitrogen gas at `STP` is nearly `3.5 R`.Now when the - YouTube
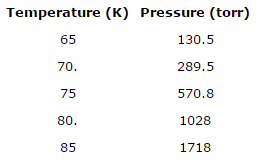
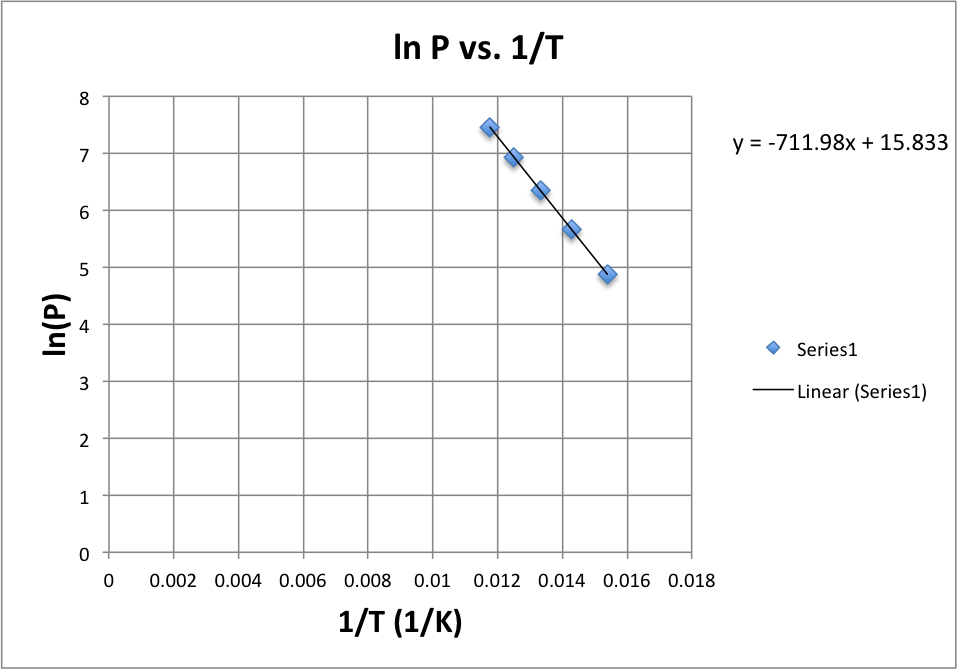
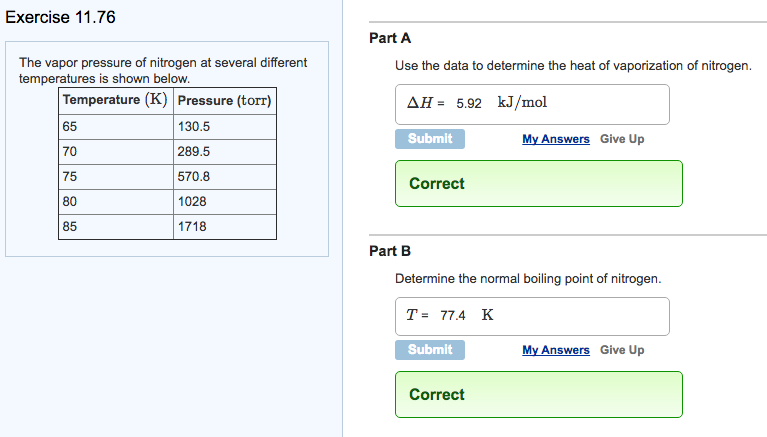
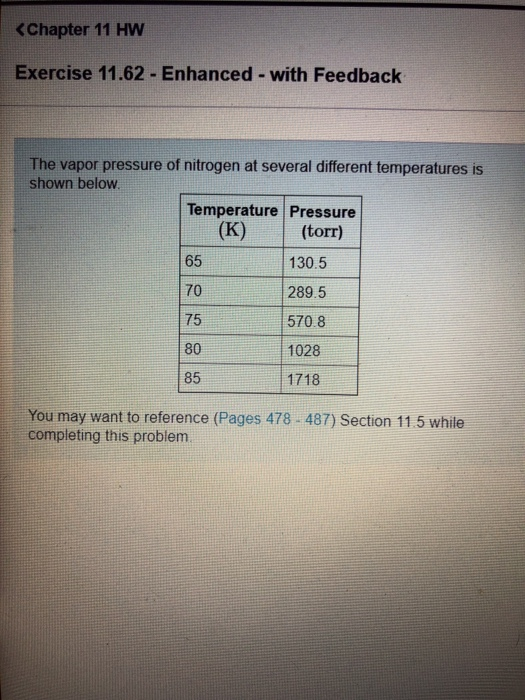
![SOLVED: Using the Clausius-Clapeyron equation O iie data provided in %he problem to graphically determine (on excc] spread shect) the heat of vaporization Of nitrogen. Graph the data below manner used detenmnine SOLVED: Using the Clausius-Clapeyron equation O iie data provided in %he problem to graphically determine (on excc] spread shect) the heat of vaporization Of nitrogen. Graph the data below manner used detenmnine](https://cdn.numerade.com/ask_images/be895267f44e437c8bb031c3001bb014.jpg)
SOLVED: Using the Clausius-Clapeyron equation O iie data provided in %he problem to graphically determine (on excc] spread shect) the heat of vaporization Of nitrogen. Graph the data below manner used detenmnine

Table 1 from Molar Heat Capacity (Cv) for Saturated and Compressed Liquid and Vapor Nitrogen from 65 to 300 K at Pressures to 35 MPa | Semantic Scholar

SOLVED: 3. The latent heat of vaporization of liquid nitrogen is about 200 kJ/kg. Suppose u have 1.00 kg of liquid nitrogen boiling @ 77.0 K. If u apply heat at a

![Latent heat of vaporization for main components of LNG [10]. | Download Table Latent heat of vaporization for main components of LNG [10]. | Download Table](https://www.researchgate.net/publication/330572654/figure/tbl3/AS:718422421803010@1548296661881/Latent-heat-of-vaporization-for-main-components-of-LNG-10.png)