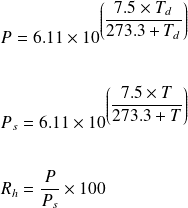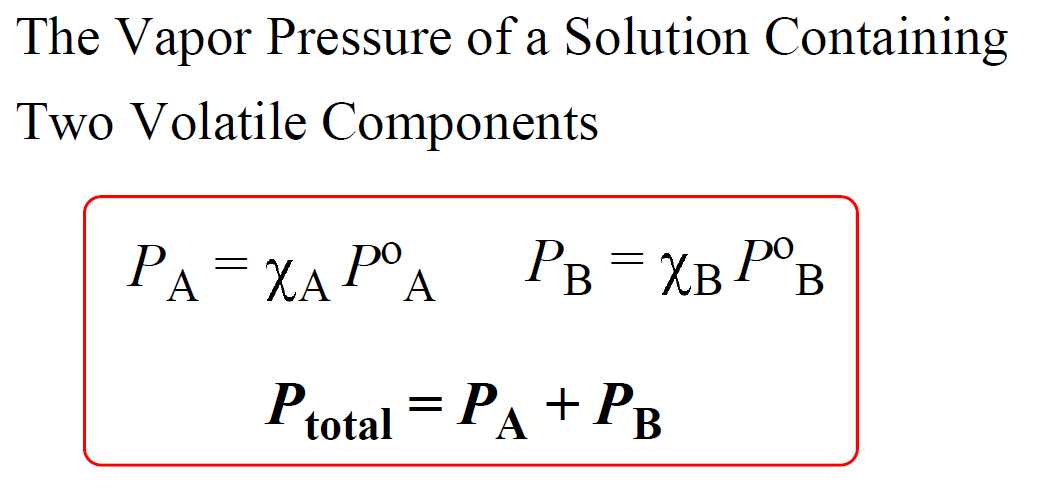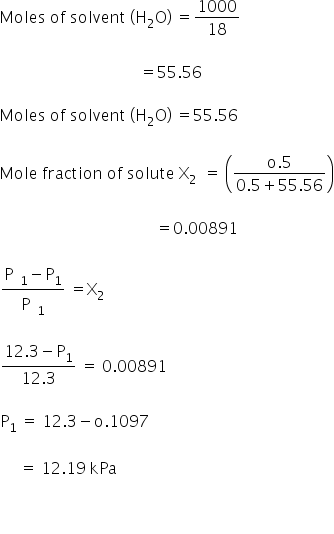The vapour pressure of a pure liquid A is 70 torr at 300 K. It forms an ideal solution with another liquid B. The mole fraction of B in the solution is
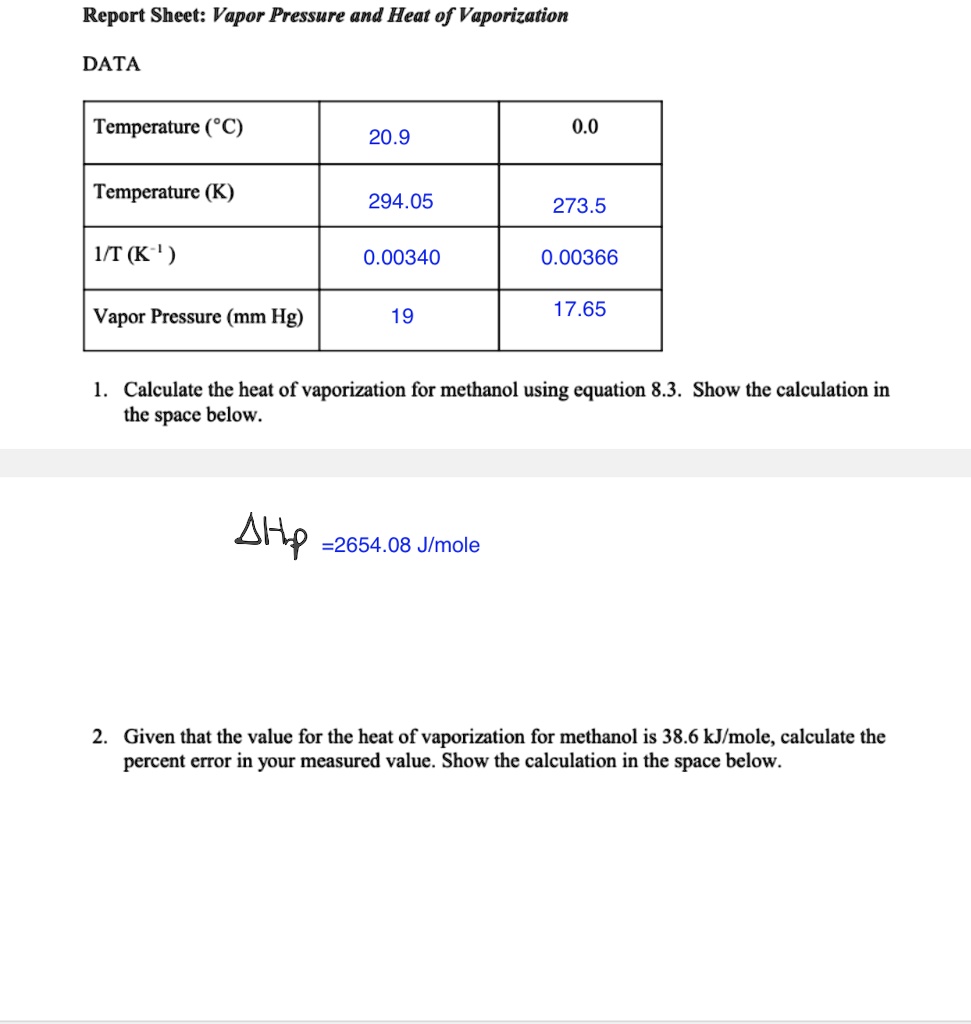
SOLVED: Report Sheet: Vapor Pressure and Heat of Vaporization DATA Temperature (*C) 20.9 0.0 Temperature (K) 294.05 273.5 IT (K ' ) 0.00340 0.00366 Vapor Pressure (mm Hg) 19 17.65 Calculate the