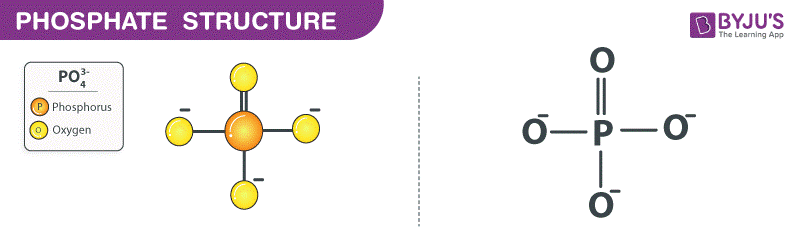
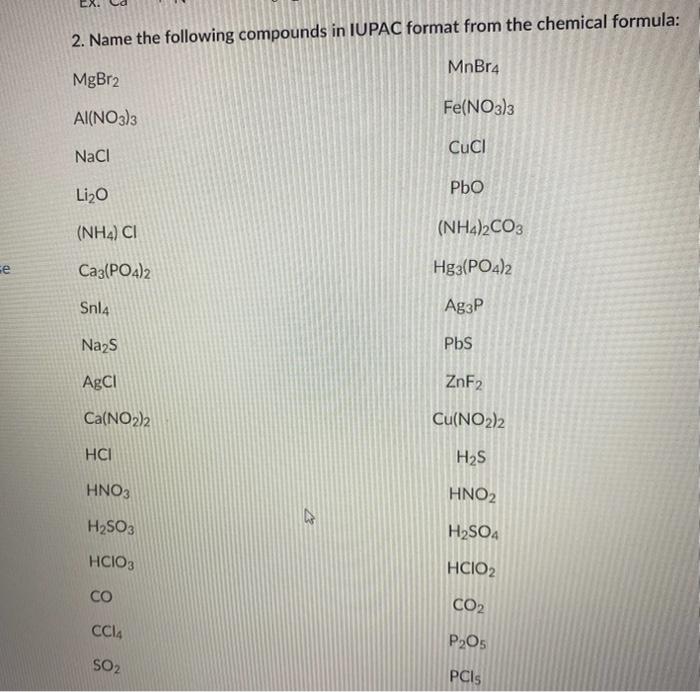
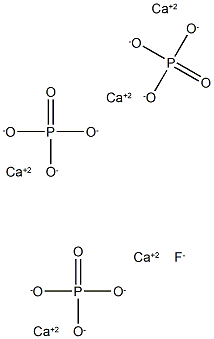
Write the chemical names of the following compounds: (a)K(2)SO(4) (b)Mg(3)(PO(4))(2) (c)NH(3)Cl (d)ZnS (E)Na(2)N (f)AgBr

SOLVED: The name of the compound with formula Fe3 (PO4)2 is: Iron (III) carbonate Iron (II) Phosphate Iron (II) Oxide Iron (III) Phosphate

Naming Ionic Compounds Chemistry Dr. May Ionic Compounds Formed when electrons are transferred from the less electronegative atom (Na) to the more. - ppt download

SOLVED: Predict the chemical formula for the ionic compound formed by (a) Ca2+ and Br-, (b) K+ and CO3 2-, (c) Al3+ and CH3COO-, (d) NH4 + and SO4 2-, (e) Mg2+ and PO4 3-.

Question Video: Recalling the Chemical Formulas for the Calcium Compounds Used in Fertilizers | Nagwa

Ternary Compounds Ternary compounds are those containing three different elements. (NaNO3, NH4Cl, etc.). The naming of ternary compounds involves the. - ppt download