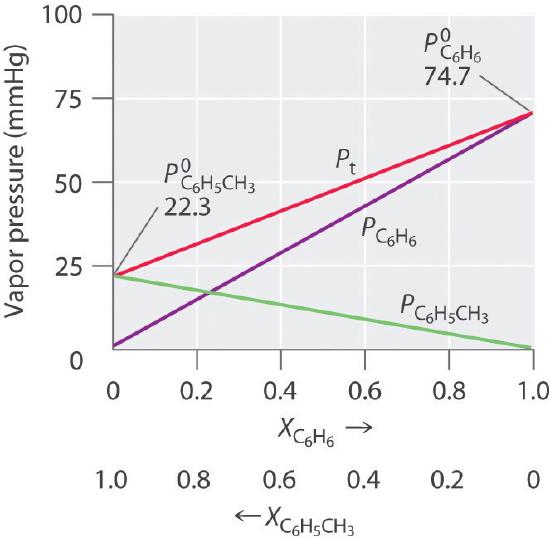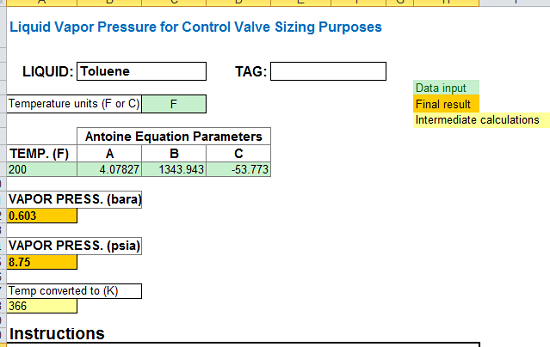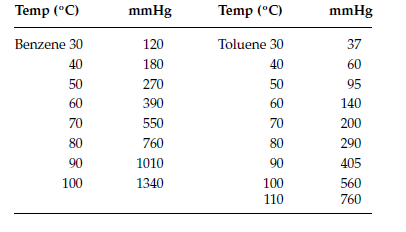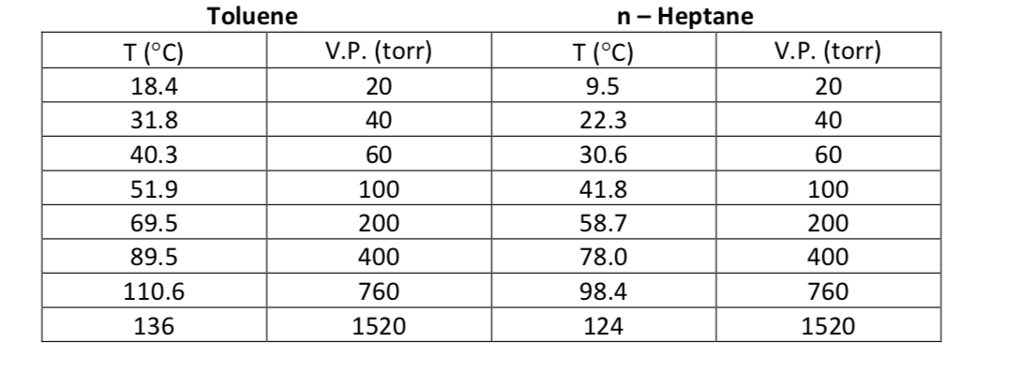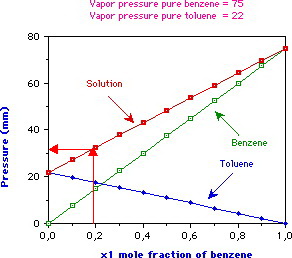
Toluene and benzene have vapour pressures at 20 °C of approximately 22 mmHg and 75 mmHg, respectively. Calculate the total pressure of an ideal solution made up of 0.828 mol toluene and
![Vapor pressure of benzene. Line: Experimental values [34]. Circles:... | Download Scientific Diagram Vapor pressure of benzene. Line: Experimental values [34]. Circles:... | Download Scientific Diagram](https://www.researchgate.net/publication/230880787/figure/fig3/AS:669250792472583@1536573231374/Vapor-pressure-of-benzene-Line-Experimental-values-34-Circles-inhomogeneous-MC.png)
Vapor pressure of benzene. Line: Experimental values [34]. Circles:... | Download Scientific Diagram

The vapour pressure of pure benzene at 88^oC is 957 mm and that of toluene at the same temperature is 379.5 mm. Calculate the composition of benzene - toluene mixture boiling at 88^oC .

Benzene and toluene form an ideal solution over the entire range of composition. The vapour pressure of pure benzene and toluene at 300 K are 50.71 mm Hg and 32.06 mm Hg