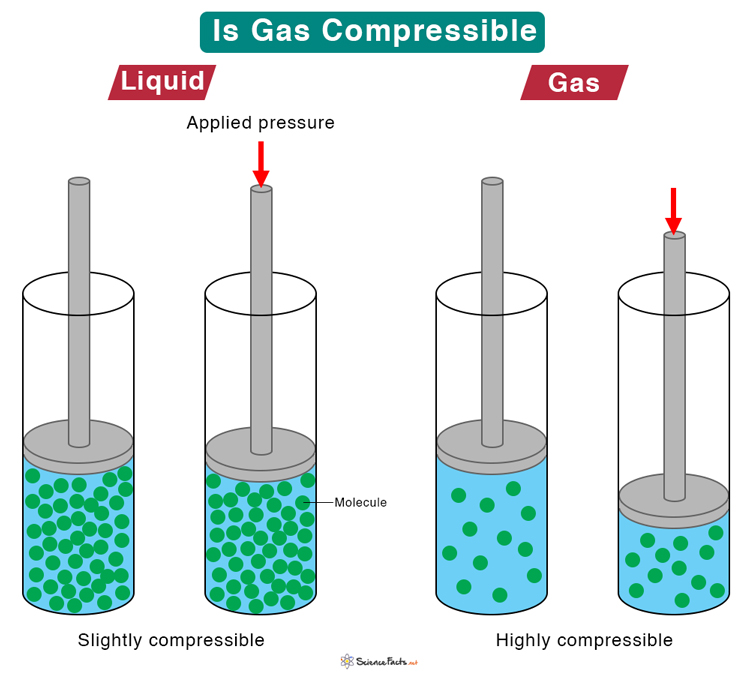
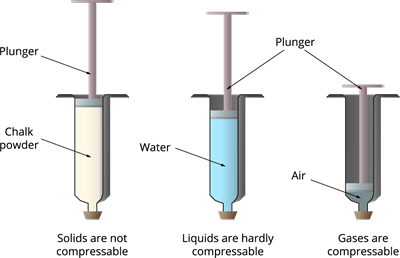
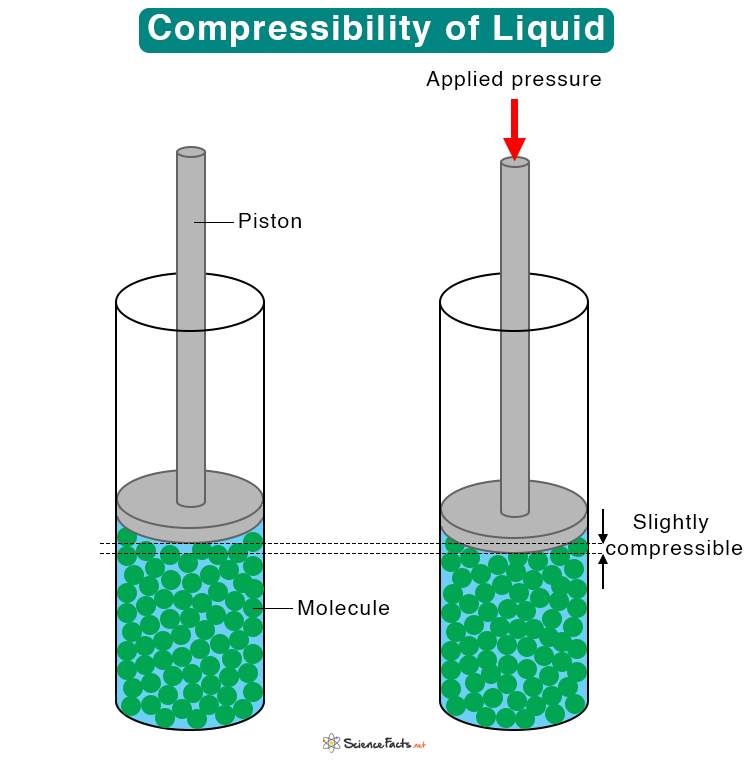
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas

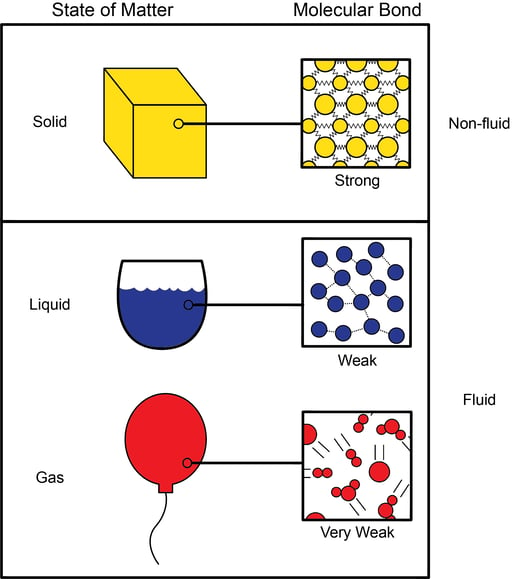
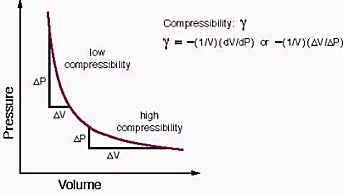
Aim: What are the properties of Gases?. Compressibility Compressibility is measure of how much volume decreases under increased pressure. Gases are easily. - ppt download
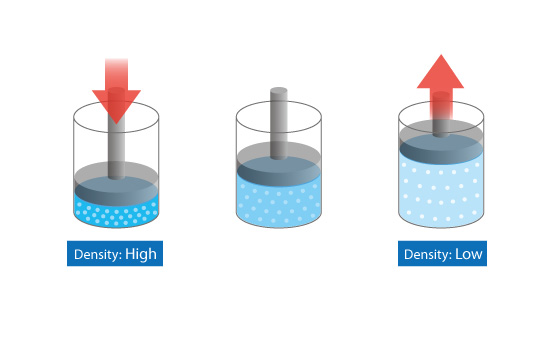
Basic Course of Thermo-Fluid Analysis 06: Chapter 3 Basics of Flow - 3.2.1 Compressible/incompressible fluids|List

1 Behavior of Gases Ch Why do air bags work? Which would you rather hit the dashboard or an air bag? Why? Which would you rather hit the dashboard. - ppt download

Compressibility factor, Z of a gas is given as Z = pV/nRT (i) What is the value of Z for an ideal gas?(ii) For real gas what will be the effect on